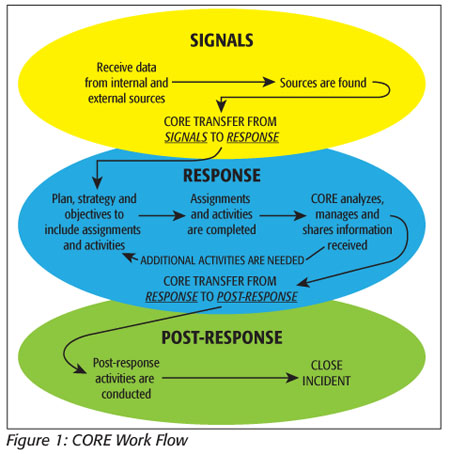
Every second counts. Never is that more true than when dealing with illness outbreaks. A faster disease outbreak response can mean fewer illnesses. With that in mind, the U.S. Food and Drug Administration (FDA) set about creating an organization that would accelerate and streamline the agency’s foodborne illness response, and a year ago this month, when the pieces were put in place to create the Coordinated Outbreak Response and Evaluation (CORE) Network, it was ready to test this new concept (Figure 1).
 In the past, upon learning of an outbreak, FDA would form a team, drawing staff from across the agency to respond. With CORE, FDA made a commitment of full-time staff dedicated not just to coordinating the response to an outbreak that was underway, but also to more rapidly identifying new outbreaks, and developing plans and policies that might prevent outbreaks in the future.
In the past, upon learning of an outbreak, FDA would form a team, drawing staff from across the agency to respond. With CORE, FDA made a commitment of full-time staff dedicated not just to coordinating the response to an outbreak that was underway, but also to more rapidly identifying new outbreaks, and developing plans and policies that might prevent outbreaks in the future.
CORE divides outbreak response activities into three phases:
• Phase One: Find the outbreak
• Phase Two: Stop the outbreak
• Phase Three: Prevent the next outbreak
By assigning teams of experts to each of these phases, FDA expected to accomplish three goals. FDA expected to respond with increased speed. It expected to respond with increased consistency, and it expected to incorporate lessons learned from each response into future responses and into policies and other actions aimed at preventing future foodborne illnesses.
“Our teams include epidemiologists, microbiologists, veterinarians, environmental health specialists, consumer safety officers, and policy analysts,” explains Dr. Kathleen Gensheimer, chief medical officer and director of CORE. “By dedicating experienced staff from a range of pertinent fields to each phase, we create a consistent, continuous operation. I think this has been key to the efficiency and success of this three-pronged effort to fight foodborne illness.”
Experience—a CORE Competency
In staffing CORE, FDA recruited from its pool of experts who had previously responded to outbreaks, therefore ensuring that hard-won experience was the very foundation of the new system. From the beginning, nobody in FDA doubted the concept, but certainly nobody could have predicted it would be tested and proven to work the very next month. Yet on the very day in 2011 that FDA officially announced in a press release that CORE was operational, the agency was issuing another news release warning consumers of what would become the deadliest outbreak of foodborne illness in decades.
During that outbreak, linked to listeriosis in fresh, whole cantaloupe from Jensen Farms in Colorado, CORE would test its processes under fire and validate the underlying concept of its creation. But more importantly, through those processes, CORE would save lives.
“It all starts with signals,” says Jeffrey Brown, head of CORE’s Signals and Surveillance Team.
Brown’s team searches through information on FDA-regulated products that is reported into various databases by local and state health agencies, through news stories, and through information shared by FDA field offices. The team members are looking for signals or “red flags” that could be an early warning of an emerging outbreak.
They are in frequent contact with the U.S. Centers for Disease Control and Prevention (CDC), sharing information about food products, disease surveillance trends, and cases of illness occurring in various parts of the country. In addition, the Signals Team searches FDA data for historical information on firms, such as past inspections or sampling results, all in an effort to “connect the dots.” The team compiles this information into a concise package that will allow the assigned CORE Response Team to hit the ground running.
CORE at Work: Trial by Fire
During last year’s listeriosis outbreak, which made 146 people ill and killed 30, the Signals Team was able to provide information garnered from Internet research showing that Jensen Farms had not yet completed harvesting cantaloupe in the region of the country where many illnesses were being reported.
From there, the CORE Response Team took over in pursuit of their single-minded goal—stop the outbreak. The information passed on from the Signals Team to the Response Team allowed quick mobilization of FDA investigators, who, along with key partners like CDC and Colorado state officials, inspected Jensen Farms and were able to collect samples confirming the farm as the source of the listeriosis outbreak. By September 14, Jensen Farms had initiated a recall of their entire 2011 production line that spanned at least 24 states.
“The response doesn’t end with a press release announcing the recall, though,” says Roberta Hammond, CORE’s response manager. “Once the recall starts, we continue coordinating efforts with those of other FDA offices to monitor the outbreak, inform the public, and ensure the effectiveness of the company’s recall.”
In monitoring the effectiveness of the recall, FDA audited nearly 100 percent of the first-tier customers receiving Jensen Farms cantaloupe and many additional secondary receivers of the product. A month after it began, FDA deemed the recall complete.
While the Response Team was still actively involved with the outbreak, the Post-Response Team coordinated a vitally important task: an environmental assessment of Jensen Farms to determine what factors might have contributed to the outbreak. The purpose of this type of assessment is to determine how the environment might have contributed to the introduction and transmission of pathogens that have caused illness.
“This was the first time we had seen listeriosis in whole cantaloupe,” says Don Kautter, CORE’s prevention manager, “so it was very important that we be able to assess the processes and conditions on the farm and determine what lessons we could take away from this tragedy.” With the cooperation of Jensen Farms, FDA, state, and local officials conducted the environmental assessment at Jensen Farms in late September.
The assessment team had expertise in produce safety, agriculture, veterinary medicine, epidemiology, environmental health, and sanitation. This team identified the following factors as those that most likely contributed to the introduction, spread, and growth of Listeria monocytogenes in the cantaloupes:
• There could have been low-level sporadic L. monocytogenes in the field where the cantaloupes were grown, which could have been introduced into the packing facility.
• A truck used to haul culled cantaloupe to a cattle operation was parked adjacent to the packing facility and could have introduced contamination into the facility.
• The packing facility’s design allowed water to pool on the floor near equipment and employee walkways.
• The packing facility floor was constructed in a manner that made it difficult to clean.
• The packing equipment was not easily cleaned and sanitized; washing and drying equipment used for cantaloupe packing was designed for and previously used for postharvest handling of another raw agricultural commodity.
• There was no precooling step to remove field heat from the cantaloupes before cold storage. As the cantaloupes cooled, there may have been condensation that promoted the growth of L. monocytogenes.
The findings of the environmental assessment have subsequently been used for educational outreach and guidance to the cantaloupe industry.
“A major goal of the Post-Response Team is to provide information to update and improve agency policy or industry guidance that can help prevent future outbreaks,” says Brett Podoski, head of CORE’s Post-Response Team. In revisiting CORE’s first outbreak response, it is important to understand the pieces that make up the CORE Network.
An Extensive Response Network
The CORE Network includes all the key strategic FDA resources in place in the field—the district offices and regional specialists; the regional emergency response coordinators that work with state partners; the Office of Crisis Management at FDA; FDA’s Offices of Public Affairs and External Relations; and the subject-matter experts at the Center for Food Safety and Applied Nutrition and the Center for Veterinary Medicine. However, the CORE Network is far more than that.
“When thinking about CORE, it is crucial to remember that CORE extends beyond FDA,” says Dr. Gensheimer. “The CORE response teams are coordinating FDA resources with the resources and efforts of agencies at every level of government.”
FDA regularly partners on outbreak response with federal agencies like the U.S. Department of Agriculture and CDC, as well as with state departments of health and agriculture and with local health and safety agencies.
Since that first test of its capability, CORE has responded to more than 40 outbreaks of varying degrees over the past year.
“Outbreaks can vary in size and in the severity of the illness, and a major challenge with some outbreaks is the scarcity of information,” says Hammond. “Sometimes there is not enough information to determine the cause, but we take each incident as far as we can.”
“Our Signals and Response Teams are typically working on several outbreaks at any moment,” says Dr. Gensheimer. “Once one outbreak is winding down, there’s usually no time before the next one. The Post-Response Team makes sure that we take what we have learned and apply it to future incidents. To me, that’s a recipe for providing the nation with a nimble, flexible capacity to more rapidly respond to, investigate, and prevent foodborne outbreaks. CORE will only get better in each coming year.”
Douglas Karas is a communications specialist with the CORE Network. He has 17 years of experience in public affairs, having worked for the U.S. Air Force and, most recently, as a press officer for FDA.
>