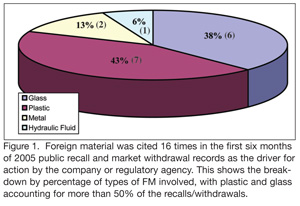
 Historically, foreign material (FM) control has been an Achilles heel for many food processors. This is because FM inclusion in finished product is one of the leading sources of consumer complaints across all sectors in the food industry. Published enforcement reports for the first six months of 2005 have presented a significant flurry of market actions related to FM inclusions. Figure 1 represents the breakdown of 2005 public recall and market withdrawal records in which FM is cited as the driver for action by the company or regulatory agency. The business impact associated with completing a market action can be drastic, and usually isn’t considered as a key element of any successful business strategy. Initiating the retrieval and disposition of just one production day of a single SKU from a national retail chain can quickly escalate to a seven-figure excursion to the bottom line. This is especially true when sales are not generated for a lengthy period of time because there is no available “good” replacement stock to place on shelves.
Historically, foreign material (FM) control has been an Achilles heel for many food processors. This is because FM inclusion in finished product is one of the leading sources of consumer complaints across all sectors in the food industry. Published enforcement reports for the first six months of 2005 have presented a significant flurry of market actions related to FM inclusions. Figure 1 represents the breakdown of 2005 public recall and market withdrawal records in which FM is cited as the driver for action by the company or regulatory agency. The business impact associated with completing a market action can be drastic, and usually isn’t considered as a key element of any successful business strategy. Initiating the retrieval and disposition of just one production day of a single SKU from a national retail chain can quickly escalate to a seven-figure excursion to the bottom line. This is especially true when sales are not generated for a lengthy period of time because there is no available “good” replacement stock to place on shelves.
With the global media’s current scope of influence on consumer perception of the safety of food products, damage to brand equity can be severe when a public announcement is made about the initiation of a market action for FM, or when coverage is focused on an isolated local event. Unlike biological and chemical hazards in foods that are largely intangible to the typical consumer, most people can conceptualize what ground glass, knife blades or hard plastic chunks would look like in a product. The reaction of many consumers to this type of inclusion is how did “they” (meaning the food manufacturer) let that happen? To illustrate this point look no further than the publicly traded corporation Wendy’s. The exoneration given to the chain for the alleged “finger-in-the-chili incident,” which was found to be a fraudulent claim for which the hoaxers are currently and rightly being prosecuted, did not help their quarterly results but it certainly gave late night talk show hosts and other media personalities a plethora of fresh material to use while dragging Wendy’s brand name through the mud.
The deleterious effects of FM inclusions in food products go beyond business results, however. When FM is consumed by an unsuspecting consumer the result can exceed the gross factor, potentially posing injurious or even deadly consequences. Because of this, all food manufacturing operations are well served by implementing proactive, comprehensive continuous improvement strategies and utilizing the best available FM control technologies to reduce the incidence of such inclusions.
Defining Foreign Material
There are many regulatory definitions of what constitutes foreign material and they should be presented prior to moving forward with the topic. My personal definition of FM is a little more simplified and broader in scope than those of the regulatory agencies. FM, referred to in various circles as foreign objects and/or extraneous matter, is defined as any material(s) whose quantity and presence within a food product is unanticipated by the customer or consumer purchasing the product and/or the regulatory body that has jurisdiction over the processing and distribution of the product. It is hoped that this definition better models the consumer and regulatory expectations for food products without getting bogged down in semantics. The use of the term “regulatory body” is inclusive of sanctioning organizations for characteristics such as organic.
The regulatory agencies define FM in a narrower scope than the definition above, but it is crucial that you explicitly understand these to ensure organizational decision making processes are based on a solid base of knowledge. The U.S. Food and Drug Administration (FDA) has adopted a broad definition of FM that is quite definitive in scope. Within the Federal Food, Drug, and Cosmetic Act there are three citations that are relevant in defining foreign materials from section 402(a):
• 402(a)(1), details the position that “a food shall be deemed to be adulterated if it bears or contains any poisonous or deleterious substance which may render it injurious to health.”
• 402(a)(3), states “a food shall be deemed to be adulterated if it consists in whole or in part of any filthy, putrid, or decomposed substance, or if it is otherwise unfit for food.”
• 402(a)(4), states “a food shall be deemed to be adulterated if it has been prepared, packed, or held under insanitary conditions whereby it may have become contaminated with filth, or whereby it may have been rendered injurious to health.”
The Good Manufacturing Practices (GMPs) also make reference to FM. Section 110.80(b)(8) of the GMPs states that: “Effective measures shall be taken to protect against the inclusion of metal or other extraneous materials in food. Compliance with this requirement may be accomplished by using sieves, traps, magnets, electronic metal detectors, or other suitable effective means.”
The U.S. Department of Agriculture’s Food Safety Inspection Service (USDA FSIS) has redefined FM via issuance of FSIS Directive 7310.5- 5/30/03. Within Section V of this directive, FM is referenced to and defined as follows: “Foreign material: Foreign materials are non-animal objects, such as metal, plastic, rubber, glass, wood, steel, or lead shot.” FSIS, like the FDA, also acknowledges the presence of such materials as adulterants.
There are certain situations where FMs are considered unavoidable contaminant in food processing. The defect action levels (DALs) published by FDA’s Center for Food Safety and Applied Nutrition (CFSAN) are a great source for identifying whether the agency recognizes your specific FM of concern as an unavoidable contaminant when found below threshold levels. The DALs were created based on a minimal purity level obtainable within the confines of available technology for the processor. Since many of these DALs haven’t been updated in years, a wise processor will not even approach the maximum cited levels at any frequency. Remember, these performance standards were based on the minimum achievable level for preexisting technologies and aren’t meant as permission slips for poor operational practices or sourcing strategies.
Armed with the regulatory definitions we can now categorize FM concerns by asking key questions pertaining to whether or not the FM represents a:
1. Potential regulatory concern
2. Consumer perceivable aesthetic issue (these can sometimes still represent a regulatory concern).
3. Threat of significance to public health
Obviously, some FM will fall into more than one category and can even exist in all three categories simultaneously. Any FM in Category 3 requires priority consideration and resource appointment by the processor.
Public Health Hazards Associated with Foreign Materials
When developing a new Hazard Analysis and Critical Control Point (HACCP) plan, or when auditing an existing plan, one usually proceeds with a thorough review of the process design, ingredients utilized, human factors and other prerequisite programs to identify the sources of and adequacy of control for hazards that are significant and reasonably likely to occur. The criteria necessary to classify a FM as a physical hazard are pretty well defined by the North American regulatory agencies, and are appropriately based on a combination of science and empirical data from individual case histories. FDA defines this criteria in Chapter 5 of its Office of Regulatory Affairs (ORA) compliance policy guide issued March 23, 1999. The FDA regulatory guidance states: “The following represent the criteria for direct reference seizure to the Division of Compliance Management and Operations (HFC-210) and direct reference import detention to the districts:
(a) The product contains a hard or sharp foreign object that measures 7 mm to 25 mm in length; and
(b) the product is ready-to eat (RTE), or according to instructions or other guidance or requirements, it requires only minimum preparation steps; e.g., heating that would not eliminate, invalidate or neutralize the hazard prior to consumption.
Samples found to contain foreign objects that meet criteria (a) and (b) above should be considered adulterated within the meaning of 21 U.S.C. 342(a)(1).”
The policy further discusses how the presence of FM that doesn’t meet the above criteria could still evoke review by CFSAN to exonerate it as a hazard to public health. The FDA also has communicated that the health of certain subpopulations may be deleteriously affected by hard or sharp foreign objects between 7 mm and 2 mm (namely infants and elderly) and that they too may be viewed as a hazard in such a situation. The health hazard potential of these objects resides in the areas of potential laceration, dental injury, other gastrointestinal trauma (perforations) and/or source of infection.
USDA FSIS defined criteria for identifying FM as potentially hazardous to the public at a two-day public conference held by the agency in Omaha, NE, September 24-25, 2002. A coalition of personnel from food processing firms, academia and USDA openly discussed what criteria would dictate a FM inclusion be defined as a hazard to public health. The group concurred that the FDA standard was valid and scientifically justifiable. The officials present expressed that FSIS follows similar size criteria (7-25 mm) in determining public health hazards presented by hard or sharp FM. FSIS also eluded to the fact that they will evaluate complaints, and sampling finds on a case by case basis (just like FDA) even if the size and preparation criteria are not exceeded. As a side note it was interesting that a dual jurisdiction recall scenario occurred this year with significant quantities of a frozen egg roll product being contaminated with glass fragments. The agencies differed in their final classification of the recall (FSIS Class I vs. FDA Class II) as noted in enforcement records. Potentially, there was a significant enough difference between the meat-containing and non-meat-containing products to justify the differing taxonomy.
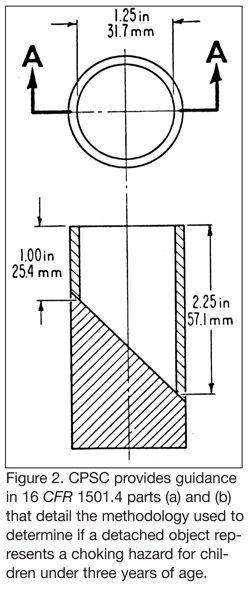 The Omaha coalition also made reference to FM as a potential choking hazard, and the relevant standards used by the U.S. Consumer Products Safety Commission (CPSC) to define such hazards. The CPSC provides guidance in 16 CFR 1501.4 parts (a) and (b) that detail the methodology used to determine if a detached object represents a choking hazard for children under three years of age. The illustration from the CFR describing the fixture used to test small parts is included as Figure 2. Though this definition is helpful in understanding how the regulatory bodies screen for potential choking hazards there is still much debate in the scientific community over what actionable criteria can be developed to classify aspiration or occlusion hazards. The FDA has, however, made a broad and sweeping statement of one product type that represented an unsuitable choking hazard for the public when it requested recall of a myriad of imported conjac gel candies in 2001 and 2002. According to public record, FDA consulted with experts from the CPSC before action was taken—unfortunately, there were several choking related incidents and a number of deaths prior to the candy being removed from the marketplace.
The Omaha coalition also made reference to FM as a potential choking hazard, and the relevant standards used by the U.S. Consumer Products Safety Commission (CPSC) to define such hazards. The CPSC provides guidance in 16 CFR 1501.4 parts (a) and (b) that detail the methodology used to determine if a detached object represents a choking hazard for children under three years of age. The illustration from the CFR describing the fixture used to test small parts is included as Figure 2. Though this definition is helpful in understanding how the regulatory bodies screen for potential choking hazards there is still much debate in the scientific community over what actionable criteria can be developed to classify aspiration or occlusion hazards. The FDA has, however, made a broad and sweeping statement of one product type that represented an unsuitable choking hazard for the public when it requested recall of a myriad of imported conjac gel candies in 2001 and 2002. According to public record, FDA consulted with experts from the CPSC before action was taken—unfortunately, there were several choking related incidents and a number of deaths prior to the candy being removed from the marketplace.
More Than a Physical Hazard
Most food safety and quality professionals understand the concept that FM may represent more than a physical hazard. Unfortunately, sometimes flawed logic is used when dealing with FM issues. Many incorrectly screen FM inclusions in food products through a single dimension of analysis, asking “Does it meet the definition of a physical hazard? If it doesn’t, then we’re not dealing with a public health situation…right?” There are other dimensions that must be considered when classifying whether an FM inclusion represents a hazard to public health; i.e., the introduction, or promotion of biological and chemical hazards either directly or through a potential interaction scenario. An example of FM as a chemical hazard can be illustrated in a scenario in which fresh produce is sourced from or staged in regions that also supply tree nuts or peanuts.
Let’s examine a hypothetical operation that utilizes such produce for making a value-added product. During the sorting step at the manufacturing facility, various plant materials, including nuts, are occasionally found to be mixed in with the produce in addition to dirt, stones and every now and then, a mammal or reptile. The personnel stationed at the manual sorting station state that at times the nuts are hard to discern due to the volume and burden of depth of produce handled in a typical production day. Is it wise for the sorting personnel to simply attempt to pick off the FM as it is discovered, or should an alternative approach be taken? The answer seems obvious due to the potential chemical hazard, but in the absence of employee education, clearly written policies, procedures and work instructions, this situation could quickly become a train wreck for the processor. The transportation company was the vector for this example because they also haul peanuts from local fields for other processors in the area without applying appropriate cleaning and sanitation measures afterward.
Examples of FM-associated biological hazards include items like bandages, exposed insulation or other friable materials that are introduced to packaging material streams that will soon house micro-sensitive RTE products. All the intervention steps taken up to that point to ensure a microbially stable product are potentially nullified by the FM inclusion.
Many people who aren’t seasoned practitioners of risk assessment and situational hazard analysis may also miss potential interaction effects that are a derivative of a FM inclusion. An interaction scenario can be defined by analyzing whether the foreign material would nullify the effectiveness of HACCP prerequisite programs or critical control points (CCPs) required to deliver a safe product to the end user. When analyzing an interaction scenario a critical approach is necessary because the outcome must be 100% corollary with the contemporary body of scientific knowledge available. A good example of an interaction scenario would be chunks of a wood pallet, or other larger foreign particle, becoming commingled with intermediate product and broken up into many pieces during a grind or milling operation. If the product is a low acid canned food (LACF) and the material is not removed by a separation or detection step prior to the filling operation, the presence of this material may violate the base assumptions of the applied thermal processing model (i.e., heat penetration, biological load, convection). This situation should bring the commercial sterility of the product into question. An even worse scenario would unfold if this type of FM were to be introduced into a continuous low acid aseptic process where the scope of microbial contamination could persist beyond the suspect batch.
Essential Aspects of FM Control Programs
The enemy of any effective FM control program is complacency by the establishment to examine the underlying business processes and systems that may lead to a serious FM incident. Naively stating that “it won’t happen to us” provides no quantitative basis for security. Vigilance in GMP compliance and the application of a thorough and scientific HACCP review are the first steps toward long-term success for your operation. Above all, however, a commitment to be proactive and to end continual firefighting scenarios must be embraced. Use of a systematic proactive approach is more useful than expending unfocused effort and capital in areas that “feel good.”
A simple process mapping exercise coupled with a hazard analysis and GMP review are far less expensive than investing in detection technologies, and many times these activities can aid in justification of the capital for such equipment if it truly is necessary. There is a place for separation and detection technologies in our industry, but these should only be employed after a thorough process and systems review.
Unless your organization is currently in turmoil with a public health or regulatory action, it is important to step above the trees and take a look at the activity within the surrounding forest. Prior to embarking upon any control scheme it is critical to understand what level of protection the status quo is providing. A retrospective analysis of the “year in complaints” can be an enlightening experience in this regard. I find a 36-month snapshot of complaint data is ideal because it allows one to identify trends in FM contamination, or to use time order series to identify seasonal influences. Unfortunately, consumer complaints are often unknowingly coded by consumer relations agencies into formats that do not afford actionable analysis by food safety professionals. A descriptor such as “Foreign Object Unrelated” attached to a complaint will mute any signal that could potentially help divine the root cause of an issue. Ideally, complaints will be broken down into categories for each FM of concern (glass, metal, wood, stones, etc.) and further delineated by geography and production area. The real power from this analysis will come from combining this data with sales volume and velocity data over a reporting interval (one month is typical) to yield a defect rate. At this point, a metric of reported complaints per million opportunities (CPMO) can be ascertained, and the scope and severity of your foreign material issues better illuminated.
As part of an overall performance benchmark, the firm can now look at other internal failure costs of FM, including quantities placed on hold, scrap generated, yield loss, incomplete orders and supplier incidents. This review process requires effort, but the holistic upstream data gathered can be an important indicator of potential for future issues. Commissioning a product purity baseline for macroscopic and microscopic FM inclusions can provide a wealth of actionable information for a processor.
Collecting and managing this information can be quite a task but possessing the data will definitely aid in conducting a hazard analysis. The data collected can also be valuable for identification of exemplary processes or practices that can be reapplied for rapid continuous improvement. I advise using both threat exposure assessment matrices (TEAM) and failure mode and effects analysis (FMEA), as quantitative tools to improve the quality of assessment, in addition to a typical HACCP systems review. FMEA is an especially good tool for augmenting your FM control systems because it allows for numerical ranking of severity, probability and control within an existing or proposed process that, when tabulated, help illustrate where resources can be better utilized.
I always have found it a lot easier to sleep at night if I know that the supply base is in alignment with our core beliefs and practices related to process control and food safety. As processors we face enough challenges in keeping FM out of food products that are within our control. Unknowingly receiving an unanticipated contaminant in a supply stream is the metaphorical equivalent of giving a drowning man an anchor. Quite simply, management of supplier expectations, facilities, GMP compliance, HACCP programs, preventative maintenance systems and GAPs are critical aspects of success. Auditing supplier locations and placing language related to FM into purchasing agreements are fundamental practices, as well. Sometimes just engaging in conversation related to FM control with a supplier can elucidate novel ideas and systems for control. Remember that partnership is key and suppliers should not be asked to do the impossible. Expectations of control and prevention must be enforced with carriers as well. Bulk carriers should be required to present wash tickets that are traceable for the current haul. Raw materials in transit are especially susceptible to external contamination and tampering. Lengthy transit routes that involve extended stoppages in non-secure facilities are especially risky scenarios. Consider tarps, serialized seals and inspection upon receipt mandatory prerequisites for supplied materials.
Internal focus is difficult to maintain in demanding production environments, but a strong resolve is necessary to ensure product safety. A focused approach to auditing is necessary to tackle FM concerns. Once the processor has identified that the basic food safety systems (GMP, GAP, PM and HACCP) are in-place, a proactive review of standard work practices, including containment (hold) scenarios, packaging handling/routing and facility/processing line condition is warranted. Some good areas to review as processes are examined are:
• Are loose items (pens, calculators, testing equipment) necessary in the production environment?
• If a thermometer, pH probe, timer or other small piece of testing equipment were lost or broken in a production area would a new one simply be supplied to the employee without question, or would there be a chain of events that would trigger a review?
• Are high pressure water or air hoses routinely used for continuous cleanup in areas where they could give FM a free “ride” into the process stream?
• In high throughput areas where ingredients are being continuously manually opened and dumped into hoppers, bins or airvey systems, what prevention exists for keeping the packaging components of the ingredient from entering the product stream (think plastic bags, metal clips from chubs, Mylar, fiber board, wood, tape)?
• Are additional lighting sources (potentially unshielded) or small mirrors ever brought into an area for equipment inspection?
• What are the established decommissioning, and subsequent commissioning, procedures for unplanned (emergency) or planned maintenance in production or storage areas (i.e., cutting, drilling, grinding, electrical wiring, hot work)?
• Are materials or products ever removed from the process flow for rework, testing and/or reconditioning? If yes, then do these products get put back into the correct point of the process so detection, separation and other controls are not bypassed?
• Are waste removal routes separated from product transportation routes?
Separation and Detection Strategies
The vast institutional knowledge of the food industry has, over time, produced many ingenuous and effective techniques to detect and separate FM from product streams. Essentially, three techniques exist for food processors and service establishments to eliminate unwanted FM: manual inspection and sorting; separation; and automated detection and rejection (which also may be manual rejection). Most processors still utilize the first method (people foraging) for given unit operations but as throughput requirements increase, most processors gravitate towards employing several of the techniques in classes 2 and 3 for added efficiency and assurance of food safety.
Separation Technologies. Separation processes exploit physical (and sometimes chemical) differences of desirable product versus FM to cull the undesired fractions from the process stream. Such exploited characteristics are inclusive of factors such as size, shape, mass, buoyancy, aerodynamic resistance and magnetic permeability. Simple questions that can be asked to aid you in your FM control program are, “Would the FM of interest float in water or an air stream? Is the FM always much larger (or smaller) in all aspects than the product? Is the FM heavier than the desired fraction?
 Typical examples of the separation processes used in our industry are sieving, screening, filtration, selective membrane technology, float tanks, pin conveyors, destoners, air classifiers and magnetic separation. Realize, however, that before a separation process is installed as a “magic bullet” one should have a good model of the degree of variability in the exploited characteristic of both the FM of concern and the product. This is important to ensure there is minimal overlap. Overlap of characteristics in separation processes can result in either additional scrap/yield loss, or unknowingly allowing the FM to be selected for in the product itself (Figure 3). I recommend consulting an experienced vendor when selecting separation equipment, and come prepared to discuss production rates in the unit operation of interest and provide a population of samples sufficient enough for robust separator design.
Typical examples of the separation processes used in our industry are sieving, screening, filtration, selective membrane technology, float tanks, pin conveyors, destoners, air classifiers and magnetic separation. Realize, however, that before a separation process is installed as a “magic bullet” one should have a good model of the degree of variability in the exploited characteristic of both the FM of concern and the product. This is important to ensure there is minimal overlap. Overlap of characteristics in separation processes can result in either additional scrap/yield loss, or unknowingly allowing the FM to be selected for in the product itself (Figure 3). I recommend consulting an experienced vendor when selecting separation equipment, and come prepared to discuss production rates in the unit operation of interest and provide a population of samples sufficient enough for robust separator design.
Magnetic Separation. One of the most underutilized strategies for eliminating a great deal of undesirable metallic FM from both wet and dry food products is the use of magnetic separators. A well placed magnetic separator can not only aid to increase the safety and purity of one’s finished products, but it can also protect costly equipment such as pumps, mills, grinders, slicers and extruders. If the FM of interest is ferromagnetic or paramagnetic (slightly reactive in a magnetic field), it is most likely a candidate for magnetic separation.
When properly installed magnetic separators will not adversely affect process throughput or yield and can last for many years if cared for and properly handled (not dropped repeatedly or used as a wheel chock for a forklift). It is crucial that someone regularly checks the separator for contamination and document/trend findings. This check can help the processor in many ways, the most important of which is to identify the presence of FM within a process stream. If a separator is left unchecked for an extended period of time it can become saturated with material and lead to the maddening problem of random product contamination (occasionally shedding some of its load).
Detection Technologies. Unlike separation processes that are 100% active (unless they are bypassed or otherwise broken), detection schemes rely upon a different method of action. In detection scenarios a characteristic difference of the FM versus the product is identified through detection devices then signals are sent to reject mechanisms for culling. The reject mechanism can be a manual process, but this has become a less common scenario with the majority of processors favoring automated means of tracking and rejection (photo eye triggers, pusher arms, reversing conveyors, air blast, etc.). The reliability of any reject or tracking mechanism within a detection loop must be routinely tested and documented to prove adequate.
Metal Detection. There is little doubt that there has been a wide proliferation of metal detection within the food industry over the last several decades. As long as the product signal and environmental factors are conducive, the metal detector is an excellent choice for culling ferrous, stainless steels and non-ferrous metals such as brass, aluminum and lead. The latest detector designs are relatively easy to operate and quite reliable compared to those of past eras (where a technician was needed practically on a daily basis for adjustment).
Microprocessor-based digital signal processing, multiple product baseline storage and automated reject tracking/verification are all outstanding features found in modern metal detectors. Realize, however, that the principle of operation for the balanced coil design (the most widely utilized type throughout the industry) has not changed since its origination, and though many of the more common interference factors have been factored into the design of the detector, each situation will need to be independently reviewed for suitability. Also, the weakest detection signal for metal detectors are at their geometric center, so be sure that any sensitivity (size) performance language within quotations or binding contracts reflect that area.
Machine Vision. The electromagnetic spectrum is a wonderful thing because modern technology has developed detectors for qualitative and quantitative measurement of most components of the visible and invisible region. Machine vision, as defined by the Automated Imaging Association (AIA), is: “The automatic acquisition and analysis of images to obtain desired data for controlling a specific activity.” The beauty of machine vision is that it is an ever-evolving technology that offers more functionality as time passes. Essentially, if the processor has enough resources, a machine vision strategy can be employed at practically any point in the process where a human being is stationed to examine objects by their color, shape or other cues (both visible and invisible to the naked eye).
Unfortunately, “sight” is not yet highly corollary with artificial intelligence. A machine vision system relies upon programming and extensive matrices of parameters to do its job. Recognition of FMs, such as grease or tree nuts, are simply tallied up as rejects by most vision systems. A human inspector (if aware), however, may actually recognize the FM as grease and shut down the process to question its origin and severity.
X-ray. I classify X-ray systems as an extension of machine vision technologies. Modern X-ray systems are becoming more affordable for food processors thanks to the seemingly never-ending improvements in the semi-conductor industry. X-ray systems are outstanding because they have applications from inspecting product traveling through process pipelines to 100% inspection of sealed product containers at mind-numbing line speeds prior to shipment. Exploitation of density related attenuation of X-rays affords for identification of an incredible spectrum of FM—even glass, rubber, wood and some high-density plastics may be isolated.
The declining cost of high-speed X-ray detection systems is becoming further appealing as revamped hardware and talented software writers from the machine visi
Fine-Tuning Your Foreign Material Control Investment




