Have you ever had a pet that has gotten sick from eating its own food? Or how about a child who has gotten sick from sampling the dog’s food from the bowl? Even if you haven’t known a dog or a child to get sick from eating pet food, this is a very common occurrence and not fun for anyone. It has been estimated by the U.S. Centers for Disease Control and Prevention that about 1,500 humans have been infected by Salmonella through pet food sources.
The U.S. Food and Drug Administration (FDA) and the U.S. Congress have taken action to try to prevent these types of scenarios. The action is known as the Current Good Manufacturing Practices, Hazard Analysis and Risk-Based Preventive Controls for Food for Animals and is part of the recent implementation of the Food Safety Modernization Act (FSMA), which was established to provide a more preventive stance for our food industry (pet and animal food included).
According to FDA, “The Food Safety Modernization Act, the most sweeping reform of our food safety laws in more than 70 years, was signed into law by President Obama on January 4, 2011. It aims to ensure the U.S. food supply is safe by shifting the focus from responding to contamination to preventing it.”
The final Preventive Controls for Animal Food rule, published in September 2015, has also since been established to “build an animal food safety system for the future that makes modern science- and risk-based preventive controls the norm across all sectors of the animal food system.” Although many throughout the industry are anxious and a bit nervous about the implications of this new rule, a majority are glad to see science and risk being brought into the new design of the food safety system for all pets and animals.
If you’re like us (and let’s face it, there’s a good chance you are), your pet’s safety is as important to you as your child’s. For this reason alone, the new guideline released in 2015 has had tremendous support. The American Pet Products Association estimates that approximately 65 percent of households in the U.S. spend over $62 billion each year on family pets.
The Animal Food Rule
The recalls for pet food and the potential human contamination risk led Congress to establish legislation to help FDA create key regulations to help identify hazards around pet food and animal food. This legislation will support pet food companies as they work to improve their food safety systems and thus lower the risk for pet food-related illnesses and the risk for human illness through pet food. However, this final rule also includes the food safety controls for other animal food. Why does regulation reference all animals instead of just pets?
Imagine a chicken farm. Because chickens must eat, what would happen if their feed was contaminated with Salmonella? What are the odds, do you think, that the Salmonella would be transferred to the chicken dinner later served? If a hen lays an egg after she eats feed that is contaminated with Salmonella, would there be a higher risk for Salmonella to the end consumer? Salmonella-contaminated chicken feed can lead to an enormous risk for various end consumers. By controlling the risk of hazards throughout the entire food chain, the overall opportunity for contamination getting into our food supply is significantly reduced.
Thankfully, there are regulations in place designed to protect animals in our food chain, our pets and fellow humans from hazards that might affect the food and feed that are consumed. The big question often heard is: “When do we have to comply?”
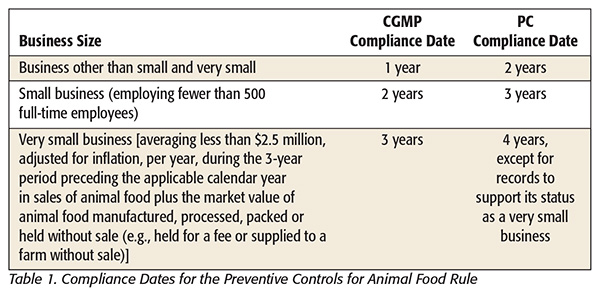 In September 2015, the final Preventive Controls for Animal Food rule was established with compliance dates dependent on company size (Table 1).
In September 2015, the final Preventive Controls for Animal Food rule was established with compliance dates dependent on company size (Table 1).
What Does It Mean?
Now, for the first time in the animal and pet food industry, there is a requirement to comply with the new Current Good Manufacturing Practices (CGMPs) for any location that must comply with the final rule mentioned above. This requirement states:
Any employees who handle, manufacture, process, pack or hold animal foods have rules and practices to follow in order to protect the products and packaging materials from contamination.
For some companies, this may be a completely new process, whereas others will just have to “tweak” their current systems to comply.
Additionally, any animal food company must ensure all its workers (seasonal and temporary) receive training in the principles of animal hygiene and animal food safety if they supervise or perform manufacturing, processing, packing or holding duties in the company. Auditors often ask the production crew making a product on a line, “What is the law or regulation requirements for making this product?” They usually receive deer-in-the-headlights looks from the workers, supervisors and managers. Most of the people could answer only basic GMP requirements like washing hands or clean clothing or not being ill when working with food. Not many individuals could fully explain how they know the products they make meet all of the law requirements or could explain how the company’s designed system ensures they meet the regulatory requirements. Could you answer that question? What about your employees?
The system is supposed to work as follows:
1. The company establishes a system of policies, Standard Operating Procedures and work instructions.
2. The people writing these documents ensure that all the requirements from the many laws (federal, state and local) as well as any other requirements (such as a Global Food Safety Initiative audit, customer requirements and company-specific requirements) are added to the policy, procedure or work instruction.
3. The company designs a training system to train the employees at the right time using an effective method and the right training message.
4. The company establishes an internal audit function to evaluate the effectiveness of the workers for following the company policies, procedures and work instructions (thus showing proof that they are following the current regulations). Usually, a finding on audits is a disconnect between what was done on the production floor and what was written in the instruction documents.
Animal food workers must know the rules for implementing food safety practices into the manufacturing, processing, packing and holding of any animal food. This doesn’t mean just when they are first hired but also periodically throughout their career. FDA expects companies to determine all refresher courses needed to implement up-to-date training schedules. Internal audits will help evaluate this process and answer the question “Is our training program working the way we need it to work?” If a company organizes its internal audit to be the most difficult audit to pass, it will find its noncompliance issues before anyone else and have an opportunity to shine during an external audit (regulatory, customer or third party).
Preventive Controls-Qualified Individuals
If you remember one thing from all your training, it’s that the policies, procedures, work instructions and GMPs should be based on the hazards identified at the facility. The processes a company implements should always be a reflection of the findings of the on-site food safety team. With FSMA, companies must design their food safety plan using an accurate facility- and process-specific Hazard Analysis, which requires a facility to identify a Preventive Controls-Qualified Individual (PCQI) for each facility.
A PCQI is required to be trained and have experience in how to effectively manufacture, process, pack and carry the products being produced. This person also must fully understand the requirements of the final Preventive Controls for Animal Food rule. The most efficient method used to show an FDA inspector that a PCQI meets all federal requirements is to produce a certificate of completion for a PCQI-approved course for animal food. This certificate is not required, but FDA inspectors accept it as evidence of meeting the requirement of the final rule. If you choose not to show the certificate, you would have to show proof of how you were trained, have the experience required to do the job and know the FSMA rule. How long would that take to show an FDA inspector?
There has been a widely used unwritten rule throughout the industry: Get the auditor in, give them what they need and get them out of the facility as quickly as possible. An FDA inspector will expect to talk to the PCQI and the person in charge while visiting the facility. He/she will probably want to see the Hazard Analysis and associated food safety plan that was performed to see what hazards exist within the facility’s processes.
Animal-Specific Hazard Analysis
While performing a Hazard Analysis for pet and animal food, it is important to fully understand the animals consuming the food, their specific nutritional needs and any possible food components that could possibly cause harm. For example, if sheep ingest too much copper with their food, they can develop copper toxicity poisoning, which is when copper builds in the liver and causes the liver cells to die and disperse copper into the bloodstream. Once this happens, sheep become lethargic and anemic, and death usually occurs within 1–2 days.
If a company makes sheep food, the amount of copper in that food becomes a potential hazard that will require an analytical process—the most critical part of the Preventive Controls for Animal Food rule. It is important to have the right people involved in the Hazard Analysis process; a cross-section of workers (production, quality, maintenance, sanitation, warehouse, purchasing, human relations, etc.) is recommended.
When identifying hazards and determining the associated risk of each hazard, don’t forget to determine which controls are already in place and successfully being implemented. During a Hazard Analysis process, be sure to properly examine the following:
1. Formulation of the animal food
2. Condition, function and design of the facility and equipment
3. All raw materials, ingredients and processing aids
4. Normal transportation practices and procedures
5. Manufacturing and processing procedures
6. Packaging and labeling activities
7. Storage and distribution procedures
8. Intended use vs. reasonably foreseeable use
9. Sanitation and employee hygiene
10. Other factors related to the food (such as seasonal issues, weather issues, humidity, etc.)
Every identified hazard must have the amount of risk determined while considering the probability and severity of the hazard. A rubric or matrix is not required, but may be found helpful, when determining this risk.
Many resources are available to help determine the probability and severity of hazards. Some resources are provided in Table 2.
The Need for Preventive Controls
Once a food safety team has properly determined and identified the hazards and risks in your process, it is time to assess whether there is a need to implement preventive controls. The final rule defines a “preventive control” as “a known or reasonably foreseeable hazard for which a person manufacturing, processing, packing or holding animal food would, based on the outcome of a Hazard Analysis (which includes an assessment of the severity of the illness or injury if the hazard were to occur and the probability that the hazard will occur in the absence of a preventive control), establish one or more preventive controls to significantly minimize or prevent the hazard in an animal food and components to manage those controls as appropriate to the animal food.”
Basically, if you don’t control the hazard, it will result in illness for the animals that consume it.
Three basic types of preventive controls are described in the final rule: process preventive controls, sanitation preventive controls and supply chain preventive controls. However, the process preventive controls will be the most often used in a pet food or animal food facility. This control is used to ensure the control of parameters (defined later) during the manufacturing or processing of the animal food.
The processes that benefit most from the implementation of any of the three preventive controls include extrusion, flushing or sequencing procedures. The process preventive controls will often include heat processing, irradiating or refrigerating animal or pet foods. The controls used here were at one time referred to as Critical Control Points (CCPs) under the Hazard Analysis and Critical Control Points system; because of this, companies may choose to keep calling them CCPs or begin using the newer term: process preventive controls.
Every preventive control established in a facility must have four components implemented and managed to be compliant with the final rule.
1. Monitoring
2. Corrective actions and corrections
3. Verification (including validation)
4. Record-keeping
Once a food safety team identifies a hazard requiring a preventive control, the PCQI should create a recall plan as part of the food safety plan; although this is not required by law, it is considered a best practice. We will discuss the recall plan later in the article.
To understand if a preventive control is successful, it is crucial to have a grasp of what renders the hazard controlled. For instance, if there is a raw meat, like chicken, being extruded and put into a kibble for a dog food, there are time and temperature constraints at which any potential Salmonella in the chicken will die and no longer be a biological hazard for the product. That time and temperature is known as a parameter (critical limit or value) for the preventive control. Once that limit is determined through research, a challenge study or a process validation, then a food safety team can establish a monitoring system to ensure that all products being extruded in the same process meet the parameter. The monitoring process lets you know you have safe products for that hazard.
If an established parameter is not achieved, the product may be unsafe.
If you have unsafe product, then the food safety team must determine the corrections and corrective actions that must be taken to ensure that no adulterated product is shipped out. These corrective actions and corrections should be in writing before the entire process begins so everyone is aware of what to do if the values are not achieved.
The final rule defines a “correction” as an action taken to identify and correct a problem that occurred during the production of animal food. A “corrective action” is an action taken to reduce the likelihood that the problem will reoccur or to evaluate the affected animal food for safety and to prevent it from entering commerce. Corrective actions might also include performing a root-cause analysis to determine the root causes of the problem so appropriate corrective actions can be implemented against them.
Preventive controls must also be verified to ensure the monitoring, corrections and corrective actions, and record-keeping practices are being carried out as designed to guarantee no adulterated product enters commerce. Verification should include certifying the parameters are being achieved as required for a safe product. The parameters must be validated to make sure they are actually controlling the hazard. For example, does 165 °F for 1 second actually reduce the particles of Salmonella in the product or process down to safe levels for our animal customers? The temperature and time (the heating process) must be validated—in addition to the equipment used in your facility. That is, can your equipment get your product to the required time and temperature?
Controls for Sanitation Hazards
Animal food and pet food facilities will often have prerequisite controls for sanitation hazards. These are designed to control the environment where ready-to-eat animal food is processed or packaged and may be exposed to environmental hazards (such as Salmonella and Listeria). The prerequisite program controls are also used for many identified supply chain hazards. This could include an ingredient or raw material that is received without being treated for a potential hazard and then added to the company’s finished product after a heat treatment step. The total risk for the hazard is placed on the incoming material since there is no additional control in place for that material or finished product. The company can choose to control the new hazard through supply chain control, placing the control with the supplier to show that the hazard was effectively controlled. Supply chain control can be either a prerequisite program or a preventive control. Either way, the identified hazard must be controlled, as it is unlawful to ship out a product that is known to be adulterated or contaminated in any way or where it might be contaminated.
Preparing a Recall Plan
As mentioned earlier, once a food safety team has identified a hazard that requires a preventive control, a recall plan becomes part of the food safety plan. If there is a risk for a hazard requiring a preventive control getting into the product, the facility must have the capability to recall that product if needed.
The recall plan should define the three classes of recall.
Class 1 — Reasonable probability of serious adverse health consequences or death
Class 2 — May cause temporary or medically reversible adverse health consequences, or where the probability of serious adverse health consequences is remote
Class 3 — Not likely to cause adverse health consequences
The recall plan should also cover team members’ roles and responsibilities, emergency contact lists for proper notification of regulators (federal, state and local as required), customers, suppliers and the public/media. There should be specific procedures for determining the lot numbers affected and the methods for determining the effectiveness of the recall throughout the recall operation. There should also be procedures to determine the disposition of the products based on the hazards identified and where the product is located. For example, class 1 recalls should not just be thrown away due to the risk for other animals going into dumpsters and consuming the affected products. These products should instead be destroyed with a completed certificate of destruction. All recalls must be properly documented, as FDA inspectors and other outside auditors will very likely review all recall documentation.
Summary
As you can see, a lot of work is involved in making sure that pet and animal food is safe for consumption. The entire supply chain is affected, and it is important to have all pieces in the process work together to produce a safe product. You do not want a pet or child sick from the food created in your company.
Jeff Strout is a food safety training consultant at Mérieux NutriSciences.
Nick Price is the associate product manager for chemistry at Mérieux NutriSciences in North America.




