For the past 26 years, the Refrigerated Foods Association (RFA), an organization of manufacturers and suppliers of prepared, refrigerated food products and their suppliers, has dedicated itself to providing members with technical, science-based information to promote food safety, share information and expand the industry. Members include manufacturers and suppliers of wet salads, refrigerated entrees and side dishes, dips, desserts, soups, and ethnic foods, as well as companies engaged in business operations related to the refrigerated foods industry.
In 2005, RFA sponsored a study under the purview of its Technical Committee to evaluate some of the novel antimicrobial ingredients used in refrigerated foods that are designed to produce safe product by reducing or eliminating foodborne pathogens and/or to extend their shelf life by preventing or reducing the growth of spoilage organisms. This challenge study, “The Evaluation of ‘Novel Antimicrobials’ in Maintaining the Safety and Shelf Life of Refrigerated Foods,” was designed to address new areas of concern for RFA’s member companies, including regulatory compliance, zero tolerance standards, food safety assurance and confusion about the applicability and/or efficacy of new novel antimicrobial preservatives against Listeria monocytogenes.
In general, the food manufacturing industry faces the challenges of meeting the demand of consumers for quality product with longer shelf life, of maintaining compliance with regulations and of assuring food safety through best practice strategies and technologies. Microbial intervention, or hurdle, strategies and technologies are important tools at the processor’s disposal to meet these challenges, as companies search for viable options that will improve their ability to prevent, control and eliminate undesirable microorganisms in their processes and product through Hazard Analysis and Critical Control Points (HACCP), Good Manufacturing Practices (GMP) and other food safety programs. One of the microbial intervention tools that appears promising in meeting these goals are novel antimicrobial preservatives, which are ingredients that when used in the formulation of a product will chemically react in such a way that the growth of pathogens such as Listeria monocytogenes, and spoilage organisms like yeasts and molds is eliminated or retarded.
While it is well documented that standard preservatives will retard the growth of certain microorganisms, their value against Listeria is not as effective as required by U.S. zero tolerance standards in both U.S. Department of Agriculture (USDA) and U.S. Food and Drug Administration (FDA) regulations, which state that if any food product has a single organism per 25 grams at a detectable level that product is considered adulterated. Additionally, new regulations pertaining to refrigerated, ready-to-eat (RTE) meat and poultry items do not allow for the use of these standard preservatives, such as sodium benzoate. Because of this issue, a number of food industry suppliers’ antimicrobial preservatives have been proposed as effective anti-listerial compounds that would aide the refrigerated foods manufacturer in preventing the rapid and prolific outgrowth of Listeria in RTE products. Since the claims made by these companies are many and confusing, the RFA membership approved a project to evaluate the anti-listerial ability of various novel antimicrobials to obtain some clear, unbiased research to help them make decisions with regard to the use of this intervention strategy in their products.
Industry and RFA Study Drivers
The increasing availability of new and improved interventions available to processors has been driven, in large part, by the implementation of several of the FDA’s and the USDA’s more recent industry guidelines, directives and proposed rules that strongly encourage or mandate the application of intervention technologies to improve food safety. FDA’s 2004 Produce Safety Action Plan, for example, identifies as part of the agency’s objectives the promotion of risk assessments to identify with industry the microbial interventions that will be most effective in produce operations. Clearly, this is of interest to refrigerated food manufacturers that use produce multi-ingredient finished products that include fruit or vegetables in the formulation.
The USDA’s Food Safety and Inspection Service (FSIS) interim final rule on the control of Listeria monocytogenes (Lm) in ready-to-eat (RTE) meat and poultry products (Directive 10,240.4) has been the primary catalyst of the surge in the development and availability of novel antimicrobial ingredients in the marketplace. Based on the results of a risk assessment that FSIS conducted with FDA that identified several refrigerated foods (both meat-containing and non-meat containing) as high-, medium-, or low-risk for Listeria contamination, the agency identified three alternative approaches to preventing or minimizing post-lethality contamination. The Listeria RTE interim final rule, published in June 2003 and effective in October 2003, provides incentives for RTE product manufacturers to use post-lethality treatments, antimicrobials ingredients at formulation and other intervention technologies to significantly reduce the risk of the presence or growth of Listeria on these products. Each alternative impacts the level of sampling that must be done, and based on which alternative the food manufacturer chooses, the amount of regulatory oversight decreases or increases accordingly.
Briefly, the three alternatives are:
• Alternative 1: Requires the use of (1) a post-lethality treatment, which may be the use of an antimicrobial agent, that reduces or eliminates microorganisms on the product, and (2) an antimicrobial agent or process that suppresses or limits the growth of Lm. The effectiveness of (1) must be documented in the Hazard Analysis and Critical Control Points (HACCP) plan; the effectiveness of (2) must be documented in the HACCP plan or Sanitation Standard Operating Procedures (SSOPs).
• Alternative 2: Requires the use of method (1) or (2) above (same effectiveness documentation requirements apply). If option (2) under this alternative is used, the sanitation program must provide for testing of food contact surfaces for Lm or indicator organisms.
• Alternative 3: Involves the use of sanitation measures alone. A food contact surface testing program, as described under Alternative 2, is required for plants using this option.
Alternatives 1 and 2 support the use of an antimicrobial ingredient as an intervention step, and operations opting to use one of these alternatives will be subject to less USDA FSIS inspection oversight and testing. Plants using Alternative 2 will be subject to more FSIS testing than those using Alternative 1, but less than those using Alternative 3. Within Alternative 2, plants employing option 2 will be subject to more FSIS testing than those using option 1. Of course, plants using Alternative 3 will be subject to the most FSIS testing and routinely sampled on a monthly basis for the presence of Listeria, Alternative 3, therefore, places the greatest amount of pressure on the plant, since any positive finding means that product will be held and a recall will be triggered due to zero tolerance.
The majority of RFA members are currently using Alternative 3, sanitation measures alone, but since plants using this alternative are subject to the most FSIS testing, the association recommends and document the compounds to use in Alternative 1 or 2. Therefore, the project detailed here was designed to determine, if in fact, some of the commercially available novel ingredients reduce or eliminate the microorganisms, and suppresses or limits the growth of Listeria monocytogenes (Alternative 1), or if it just provides either of the above (Alternative 2).
Project Overview
Several preservative systems from three manufacturers were evaluated for their bactericidal (option 1) and bacteria-static (option 2) effectiveness against Listeria monocytogenes, and the bacteria-static effectiveness against lactobacilli and yeast (spoilage). Three different commercially prepared food products (over a wide pH range)—chicken salad, tapioca pudding and vegetable soup—were inoculated with both a Listeria cocktail and lactobacilli and yeast and the bactericidal and bacteria-static effectiveness of the antimicrobial ingredients was assessed over 35 days at 45F (±2°).
Since Listeria monocytogenes is known to grow in foods even at refrigerated temperatures which typically defeats the growth of other known foodborne illness causing pathogens—and can grow with veracity and rapidity—refrigerated temperature alone is not a single-hurdle solution. To gain real-world insight, the study was designed to keep samples at the legal holding temperature of refrigerated food (45F), rather than a colder temperature that would relegate growth, if any, of the organism to an extremely slow rate, even when inoculated. This temperature with the ±2 degrees provided researchers with temperature variation to encourage the rapid growth of the target organisms in the 35-day period.
Materials and Methods. The Listeria inoculums used in the RFA study included Listeria monocytogenes—ATCC 49594 (serotype 1/2a); ATCC 19115 (serotype 4B); and ATCC 43256 (serotype 1/2b). The spoilage inoculums used were Saccharomyces cervisiae—ATCC 9080 and Lactobacillus plantarum—ATCC 8014.
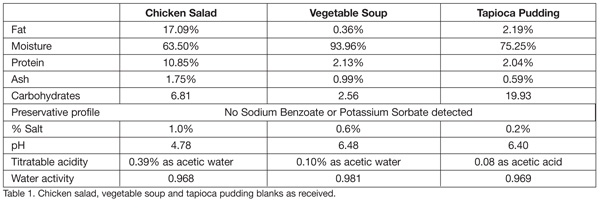 The commercially prepared, preservative-free chicken salad, vegetable soup and tapioca pudding blanks were analyzed to ensure that the range of pHs and other compositional measurements that would skew results (Table 1).
The commercially prepared, preservative-free chicken salad, vegetable soup and tapioca pudding blanks were analyzed to ensure that the range of pHs and other compositional measurements that would skew results (Table 1).
Total plate count, yeast, lactobacillus and Listeria counts were also measured on Day 0 to ensure that each sample was microbiologically appropriate. Total plate count for the chicken salad was 200 CFU/g; for the vegetable soup, <10 CFU/g; and for the tapioca pudding, 90 CFU/g. Yeast and lactobacillus counts for all three food samples was <10 CFU/g, and all tested negative for the presence of Listeria.
Each food product was separated into the number of sub-samples needed for the various treatments. Following treatment as provided by study participants, the samples were inoculated with a three-strain cocktail of Listeria monocytogenes and another set for the spoilage organisms. The treatment samples (both sets) were divided into nine sub-samples after inoculation. Three blanks of each food sample were retained that received neither antimicrobial treatments nor inoculums, each prepared separately from the rest of the experiment for each of the products as received.
The control for Listeria consisted of one non-treated sample each of the food analytes, which were inoculated with the Listeria cocktail. The control for spoilage consisted of one non-treated sample each of the food analytes, inoculated with Lactobacilli and yeast. To facilitate the addition of the treatment(s), they were incorporated into 20% of the product in a blender and then mixed into the balance of the sample. In addition, a generic antimicrobial preservative of potassium sorbate and sodium benzoate used at a maximum of 0.1 % in each of the food samples to measure the effectiveness of a more traditional preservative as compared with the controls. It should be noted, however, that these preservatives are not allowed in chicken salad under USDA regulations.
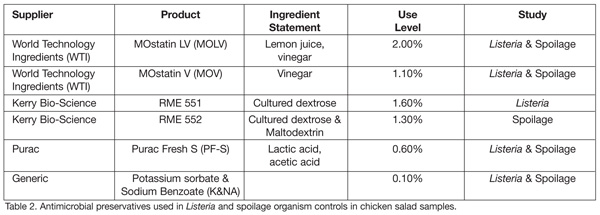 The three novel antimicrobial preservative suppliers that opted to participate in the challenge study—World Technology, Inc. (WTI), Kerry Bio-Science Americas, and Purac—each supplied two antimicrobial products to the challenge study. The product names, active antimicrobial ingredients, use level and application in the study are shown in Tables 2 and 3.
The three novel antimicrobial preservative suppliers that opted to participate in the challenge study—World Technology, Inc. (WTI), Kerry Bio-Science Americas, and Purac—each supplied two antimicrobial products to the challenge study. The product names, active antimicrobial ingredients, use level and application in the study are shown in Tables 2 and 3.

Each sample set was tested to measure bactericidal effect and bacterio-static effect throughout the 35 days on Days 0, 1, 2, 3, 7, 14, 21, 28, and 35. Each treatment type was compared by a panel of sensory analysts to the control and the blank. At each of the test days following Day 0, nine in total, the products were enumerated in duplicate for Listeria monocytogenes using Moxalactam media. In addition, when necessary, the ELISA methodology was used to determine if Listeria was present in low levels. Yeasts and Lactobacilli were also be enumerated in duplicate and the average results reported.
Challenge Study Results
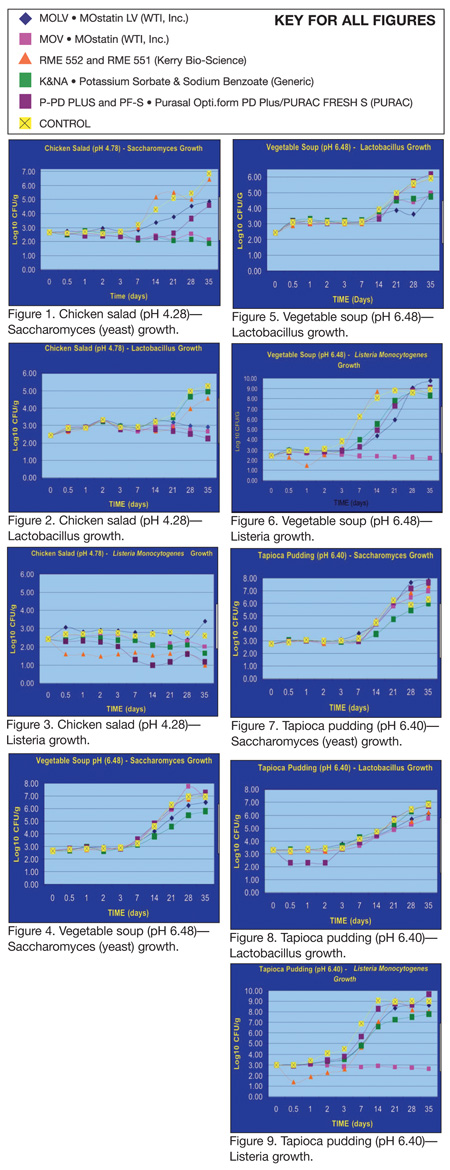 Figures 1 through 9 illustrate the results of the RFA study of the anti-listerial and shelf life effects of these antimicrobial ingredients.
Figures 1 through 9 illustrate the results of the RFA study of the anti-listerial and shelf life effects of these antimicrobial ingredients.
Chicken Salad. As shown in Figure 1, Saccharomyces (yeast) increased over the 35-day challenge study from a 2.5 log yeast inoculation of the control on Day 0 to approximately 7 log growth on Day 35. There was very little growth of yeast in any of the samples during the first three days of the study, followed by rapid growth beginning at Day 7, certainly in the control and in some of the novel antimicrobial treated samples. The generic postassium sorbate and sodium benzoate treatment and the WTI MOV preservative proved effective, achieving a slight reduction over the 35 days and preventing any growth of the spoilage organism.
Similarly, Figure 2 shows fairly rapid growth of the Lactobacillus control over the 35 days, increasing from about 2.5 logs to more than 5 logs. In this case, the potassium sorbate and sodium benzoate was not as effective. However, three of the novel antimicrobial preservatives kept the growth of this spoilage organism down to just a 0.5 log increase at the end of the 35 days.
Figure 3 confirms other scientific studies that have shown that chicken salad does not support the growth of Listeria. A number of the preservatives had a bactericidal effect, especially one, but certainly not a tremendous reduction. And, while they all slowed down the growth and achieved some measure of log reduction, none of them eliminated the pathogen to zero. Therefore, none of the novel treatments could be validated with regard to reducing Listeria to non-hazard level and certainly not to a regulatory zero tolerance compliance level.
Vegetable Soup. Due to this product’s higher pH, none of the novel ingredients performed particularly well in reducing yeast or lactobacilli growth (Figures 4 and 5). In the case of lactobacillus, although a slight log reduction by some of the novel treatments was achieved the control increased from the 2.5 logs to over 6 logs in the 35 days. Ultimately, all of the samples were at spoilage level.
One antimicrobial ingredient (WTI MOV) was shown effective in preventing growth of Listeria in the vegetable soup sample (Figure 6). However, it did not reduce the pathogen below the initial 2-log Listeria inoculation.
Tapioca Pudding. In this sample, the control for yeast grew from 3 logs to more than 6 logs over the 35 days (Figure 7). Only the traditional preservative, potassium sorbate and sodium benzoate, had any impact on reducing the spoilage organism in tapioca pudding. It is likely that the treatments of a product at this pH level appear to have encouraged this growth and that may be so due to the interaction with other organisms.
Similarly, with the Lactobacillus control, little log reduction was recorded, and for all intents and purposes, none of the antimicrobial preservatives in the study reduced lactobacilli in tapioca pudding (Figure 8).
As shown in Figure 9, only one novel antimicrobial ingredient was successful against Listeria growth during the 35 days.
For More Information
The full 75-page study is available to RFA members at no charge and to non-members for a fee. Interested parties can find the report and other technical information on the refrigerated foods industry at www.refrigeratedfoods.org.
Martin Mitchell has been the Technical Director of the Refrigerated Foods Association (RFA), the leading international association of manufacturers and suppliers of refrigerated foods, for 25 years. Mitchell is also the managing director of Certified Laboratories, Inc. with locations in New York and California. They are ISO certified and an independent laboratory specializing in microbiological and chemical analyses of food, beverages, pharmaceuticals, cosmetics and other products. His technical expertise includes all aspects of food processing, sanitation, technology and equipment. Mitchell is a recognized HACCP trainer and expert in HACCP plan design for seafood, meat, poultry, spice, herbs and refrigerated foods. He serves as director of the National Coalition of Food Importers Association, former chairman of the Microbiological Analytical Chemistry Section of American Council of Independent Laboratories and and is a member of American Spice Trade Association Executive Committee of the Technical Group. He is a member of the Food Safety Magazine Editorial Advisory Board.




