Each year, most food manufacturing plants undergo a painful and time-consuming process that results in long hours at the facility and strained relationships. No, I’m not talking about the budgeting process, although that comes in a close second. I’m talking about the annual third-party food plant audit.
These audits, though necessary to ensure that your plant’s food safety and quality assurance (QA) processes are working properly, are time-consuming and frustrating. Plant personnel are redirected from their routine duties to prepare for the evaluation. If the audit identifies deficiencies in your food safety and quality systems, the plant is required to take corrective action(s) and further, to verify that those corrective actions have solved the problem.
If you are lucky, your plant may only have to go through one audit per year. However, I know one plant that undergoes an audit nearly every two weeks because its customer base requires it. While this could be a tremendous drain on its time and resources, the company has found a way to make their audit process efficient and less painful, despite the fact that their plant is more than 100 years old! Their secret? The right amount of preparation, coupled with a positive attitude, are the main keys to establishing successful auditing practices and results. Understanding why the audit is an important element in a successful total food protection and quality system, how and when audits should be done, and what common pitfalls the food plant can avoid during an audit are fundamental to achieving improvements in your manufacturing operation’s processes and efficiencies.
Why Do We Audit and Who Should Be Doing It?
I recently posed the question, “Why do we audit?” to our plant and quality assurance (QA) managers. I received some very interesting responses, including: “Our customers require it,” “The regulatory agencies require it,” and “You make us do it.” All of these responses are technically correct; however, they overlook a fundamental reason for auditing: We audit to assure continuous improvement of our food safety and QA processes. During the past three audit seasons, Specialty Brands has reinforced this basic tenet within each of our manufacturing facilities. To accomplish this, we conduct a series of self-assessments and third-party audits, which together form an overall auditing approach that provides assurance that we are in conformance with our internal food safety and QA programs to provide our customers with safe, quality product every time.
Self-audits are vital in providing opportunities for improvement and preparation for efficient third-party audits. Specialty Brands’ philosophy is that there is no one who is tougher on us than us. Our in-plant self-assessments provide us with information on areas in which we require improvement. We have developed a tiered approach to the audit process to get the most out of the experience. First, we begin with an internal, scored self-assessment that is broken down into the following categories:
• Pest Control
• Sanitation
• Facility Conditions
• Good Manufacturing Practices (GMPs)
• Product Protection
• Material Trace
• Incoming Material
• Storage and Shipping
• Product Quality
• Security
• Continuous and Process Control
We considered using a non-scored assessment; however, we believe that scoring is preferred by our plants as it provides them with a numerical value against which they can measure themselves and their improvement. In addition, we spend time training the plant QA managers and operations managers to read, know and understand the internal assessment and third-party audit requirements for each of these audit categories.
On a monthly basis, each plant takes a section of the assessment and verifies facility conformance. The plant takes corrective action on the assessment findings. Then, at the beginning of each calendar year, each plant conducts a full assessment using the internal assessment form. This provides the plant with an opportunity to take action on deviations so that they are corrected in a timely manner prior to a third-party audit. Two of our more creative QA managers, located in the same state, took the opportunity to conduct this annual assessment at each other’s plants. This was an excellent opportunity for them to gain experience and provide “another set of eyes” in each facility.
Within a couple of months following the self-audit, a third-party auditor should be scheduled to visit the plant with the objective of evaluating performance against the corrective action plan and against the assessment plan itself prior to the actual third-party audit. This format allows the plant to practice preparing for the third-party audit, which serves as an objective measure of the company’s self-assessment efforts and whether the plants are achieving continuous improvement of its food safety and QA processes.
Most of our customers that require third-party audits provide a list of third-party auditing firms that they recognize and that can audit to their unique requirements. Processors should select an auditing service from these customer preference lists that will also provide the best service to the processsor. As you are selecting audit companies, it is important to research the specific auditors that will be utilized in your facilities. This research will ensure that the auditors’ qualifications and background are applicable to your operation. The auditor should have, at minimum, a basic working knowledge of your food category and process in order to conduct a thorough assessment, which will be more conducive to a positive learning experience for all parties involved. Ideally, the company’s relationship with a third-party auditing firm should be a close partnership in which the auditor acts as an extension of the food company’s facilities and can provide plants with honest feedback to use as improvement opportunities. As noted, also look for auditing firms that can conduct an assessment of your facilities each year after the self-assessment and prior to the third party audit.
A Third-Party Audit Template
With the self-assessment and pre-audit visit completed, your plant is now ready for the real thing: the third-party audit. Specialty Brands has found that preparation for the audit and audit flow is almost as important as the condition of the facility and documentation and thus, we try to control all facets of the audit. For example, where possible, each plant has a designated conference room or office provided for the auditor to use during the document review to afford sufficient workspace in an area where the auditor will not be disturbed. Prior to the audit, each facility participant is trained to know where to find information to satisfy each audit element. The participants organize the programs, procedures, manuals and records according to the order the auditor/assessor will review them and bring these materials into the designated audit area. This efficiency provides the best use of the auditor’s time and gives the initial impression of organization and professionalism. As is often the case, first impressions are lasting throughout the entire audit process.
Upon the auditor’s arrival, it is important that he meet the entire management team. Too often, facility audits are referred to as the “QA audit” and the burden of the entire audit falls on the QA manager. It is imperative that, at a minimum, the plant manager is present with the QA manager for the initial introduction and pre-audit meeting, as well as for the audit process and exit interview. Ideally, the meeting will include all department heads or cross-functional associates participating in the audit. This is another opportunity to present a first impression that says, “We take this process seriously.”
During the pre-audit meeting, it is important to discuss any scheduling considerations for the auditee or the auditor. For example, if the auditor wants to meet with the sanitation manager, it may be necessary to conduct the evaluation of that section first before the sanitation manager departs (since they typically work a third shift). Additionally, if a mock recall will be conducted during the audit, the time should be set and communicated so that the mock exercise can occur during the audit. At this time the auditor also can be shown restroom locations, fax availability, phone locations and power outlets for laptop and Internet connections. This is also a good time to establish lunch plans, whether the auditor would prefer to go out for lunch or to bring in food for a “working lunch” to expedite the process.
Many times the audit process begins with a “desk” audit in which the auditor will review plant programs, procedures and documents. Again, we have found that having the manuals and documents available in the conference room or office allows the desk audit to operate efficiently. After an entire day of this, the auditor should have the impression that things at this plant are organized and under control. In the event that a deficiency is noted during document review, it is entirely likely that it will be looked at less critically than if there is a feeling that the plant is well organized and in control of the situation than if things were unorganized and the plant personnel spent too much time looking for items to answer questions on the elements.
The QA manager should not leave the side of the auditor for the duration of the audit. You need to keep the audit process moving at all times. For this reason, the plant may want to use an associate as a runner. If there is a need to get additional information or records to satisfy questions from the auditor, the runner should be utilized to allow the QA manager to remain with the auditor to keep the audit moving forward. Ask the auditor if you could go on to the next item while the information is retrieved. You do not want the auditor/assessor sitting alone in a room full of your programs and documents with extra time to review things that may not have necessarily been looked at if the time had not presented itself.
The walk-through of the facility usually is next on the agenda. The plant walk-through will include the auditor, QA manager, plant or production manager, and maintenance and sanitation personnel to help answer any questions that may arise during the audit. Prior to entering the processing areas make sure that you review the plant’s GMP policies with the auditor (even if they have been in your facility before) and ask them to sign an acknowledgment and if required, a confidentiality or non-disclosure agreement. These should be re-signed at least annually. Provide the auditor with the appropriate (clean) smock, hair covering (beard cover, if needed), shoe cover, if available and sound protection, if required.
Plant management will have a pre-defined route for the audit walk-through that will move the auditor from cooked product areas back to raw product areas (reverse product flow). The plant audit team should maintain control of the audit—to a point. In fact, the plant should make arrangements to have an “advance team” move along the pre-arranged route just ahead (and out of sight) of the auditor to remove any items from the path of the auditor that either may have been overlooked or just occurred (as is likely in a real-world production environment). Where possible, any findings or comments made by the auditor during the course of the walk-through should be immediately addressed. When these situations are taken care of, it should be communicated to the auditor. If nothing else, this will let the auditor know that the plant is responsive to food safety and/or quality deficiency resolution.
The auditor’s main focus during the walk-through should be direct or potential product contamination or adulteration. He or she also will be reviewing the GMPs of the plant associates, the pest control station conditions and position, the points in the process where CCPs are monitored, and the physical conditions of the plant and surrounding grounds. The auditor will be making the determination if the plant is doing what is written in its food safety program manuals. Additionally, the auditor may ask the responsible personnel questions regarding food safety and/or quality procedures at key process areas to establish level of competency and training.
After the walk-through is complete, all plant personnel who were present for the opening meeting should be present for a closing meeting. This is another opportunity for the plant to demonstrate that it takes the audit process very seriously across all departments. The auditor should cover any deficiencies noted during the audit and the plant will verify that the auditor has identified all deficiencies noted such that there will be no surprises when the audit report arrives. Plant management personnel will indicate that they will establish corrective action timelines for any major or critical items noted by the auditor and that the plant will do everything within reason to achieve higher food safety/quality standards.
The 10 Most Common Plant Audit Pitfalls
Even the best preparation and facility conditions will invariably result in some “non-conformance” findings during an audit. Here is a “Top 10” list compiled from our experiences with common errors made by facilities and what other food companies have reported to be troublesome, with suggestions about how to avoid making the same mistakes:
1. Lack of or Inadequate Self-assessment Program. Self-audits are vital in providing opportunities for improvement and preparation for efficient third-party audits. They allow facilities to identify deficiencies in facility condition or in the plant food safety and quality assurance programs. Identification of these deficiencies should lead to corrections that will not only improve plant safety and quality performance, but also will improve preparation for and scoring on the third-party audits.
Lack of a self-audit program or infrequent self-assessments will result in failure to identify inadequacies in the food safety and quality systems or in the GMPs. Failure to take corrective action on findings will also result in repeat findings of inadequacies and will negatively impact audit final outcome. The most difficult part of the self-assessment program is the corrective action and follow-up. The system must be designed in a manner that it can be consistently and effectively maintained. This typically requires the involvement and support of a cross-functional facility team.
2. Lack of a Biosecurity Program. Following the events of Sept. 11, 2001, businesses in the U.S. increased their level of security to prevent terror attacks that might disrupt their business. While there have been no attacks on the food or agriculture business to date, our business must be considered a target. Lack of a biosecurity program increases plant vulnerability and most third-party audits now require an organized security program.
Basic features of an effective program include, but are not limited to, formation of a plant security team, visitor access control, new employee background checks, employee awareness training, security of maintenance and lab chemicals, secured allergens or restricted ingredients, and trailer security. The regulatory guidance material is a resource that facilities can utilize as they are developing and implementing a security program.
3. Inadequate Review of Pest Control Measures. Pest control is a basic component to operating a safe and sanitary food manufacturing environment. Common deficiency findings in the pest control section of an audit include missing or incomplete procedures, inadequate inspection documentation, incomplete or missing records for pesticide application, failure to complete a findings trend analysis and failure to react to findings. Many facilities, for example, lack corrective actions and documentation for items noted by the PCO on the inspection report, such as “tall weeds,” “a hole in the wall,” or “cafeteria that is not maintained.”
The trend analysis is a tool for tracking your pest control systems over a long period of time to identify repetitive activity. The designated pest control operator (PCO) should maintain this documentation and the responsible facility personnel should review it routinely. There are many resource materials available detailing an effective pest control program. It is wise to review these resource materials and compare your program against these requirements to assure that your program is effective.
4. Inadequate Routine Maintenance of Facility Condition. Common facility condition deficiencies can be indications of the lack of effective preventive management program. These include inadequate repair through the use of tape or string for corrections, poor welds or tack welds, and rust and peeling paint. Here again, monthly cross-functional self-assessments will identify facility deficiencies that can be corrected before the external audit gets underway. The monthly self-assessments allow these findings to be documented and targeted for timely corrections so that further deficiencies are prevented.
Maintaining the facility can have a definite impact on food safety and quality but facility improvements also can improve efficiencies which brings a financial benefit to the company. The program must be a long-term, systematic one— not something “thrown” together for the audit. The goal is a facility that provides a safe and sanitary environment for the plant personnel and for our finished products. If this is not consistently maintained, it is difficult to “dress it up” for an audit or customer visit.
5. Recall Plan in Place But No Practice Product Recovery. While companies may have a written recall program in place, not all conduct practice product recovery to test the effectiveness of the system. Inability to trace raw materials or packaging through the manufacturing process or finished product through distribution may result in the need for a company to expand the amount of product involved in a real product recall process. The most common items found during mock exercises are lack of traceability or records for rework/returned products, lack of packaging material records, and lack of records for inedible or discarded product.
For those companies that conduct practice product recoveries, we recommend conducting a trace on raw material, packaging and finished product at least once per year. It is highly recommended that you provide a summary of the practice recovery and document corrective action on deficiency findings. A common finding during an audit is the lack of detailed follow-through on findings from mock recall exercises. Most of your customers require mock exercises because these generally reveal weaknesses in the system. It is critical that you make the most of your time and follow through with what you learn as a result of a mock exercise. Your system will be more foolproof and the time you spend conducting the test and analyzing the results will pay off in the long-run in the event that your company is ever faced with an official recall situation.
6. Lack of Employee Training Programs. The lack of employee training programs indicates that personnel may not be receiving sufficient information to assure that they perform in a manner that provides personnel safety, food safety and quality assurance, and productivity. The training will include initial reviews of job expectations, including safety and quality, as well as ongoing refresher training. A common deficiency noted by food plant auditors is the lack of documentation of training. Training documentation is important to assure that there is a record of training received or training needed.
7. Excluding Quality Systems from the Audit Loop. While many third-party audits focus solely on assessing the adequacy of a plant’s food safety systems, it is important to have a review of the plant systems for assuring product quality, as well. Lack of current ingredient or product specifications, non-specific measurements or procedures, or inadequate or incomplete calibration of monitoring devices may result in variances from quality expectations. It is essential that you review the systems to make sure that you are doing what you say you are doing. By ensuring that these systems are part of the audit process you ensure that procedures are implemented effectively and consistently on all shifts.
8. Deficient Sanitation Standard Operation Procedures (SSOPs). All U.S. Department of Agriculture (USDA) inspected meat and poultry plants are required to have written SSOPs, and while these may not be mandated in other types of food processing, all food manufacturers should have written plant sanitation procedures that describe the frequency of sanitation and the procedures by which the plant will be cleaned.
As a means of assuring effective sanitation and avoiding audit deficiencies, plants will want to verify that the SSOPs include the following attributes: written SSOPs have been signed and dated; major changes in equipment, personnel, etc,. have resulted in reevaluation of written procedures; plans specify the frequency of procedures conducted, such as cleaning and inspection; pre-operational and operational sanitation records are prepared “real time”; and preoperational and operational inspection deficiencies are clearly described on plant records with documented corrective and preventive actions. The corrective actions should be tangible, defendable items that can be measured. If personnel are retrained, it is essential that the associated training records are documented.
9. Deficient Hazard Analysis and Critical Control Points (HACCP) Plan. HACCP is one of the most effective means of assuring that food safety is built into the process. Regulatory HACCP is an especially complex, scientific program that requires validation and reassessment. During audits the usual findings involve plan format errors, logic errors in design and decision making, document errors or failure to reassess the plans annually.
To assure the process is in compliance, verify flow charts to assure they include all the steps in the process (rework, return goods, equipment steps, and so on) and determine that the hazard analysis identifies all potential hazards inherent to the product or process. Supporting documentation and decision-making documents for determinations of critical limits, monitoring and verification activities and frequencies should be provided with the HACCP plan. Daily HACCP records that are incomplete, missing items such as date, times, initials and/or signatures will result in audit deviations as will failure for the plan to include appropriate written corrective actions in the event deviations are found as required in 417.3. Finally, pre-shipment reviews must be properly completed by plant personnel and include a full signature and date.
10. Failure to Correct Deficiencies. All of the above findings can be detected during regularly scheduled cross-functional self-assessments. However, it is not sufficient to simply document deficiencies; they must be corrected or addressed. All too often the plant will have adequate documentation of non-conformance, but lack documentation of corrective actions. This is very important in assuring that all safety and quality procedures are functioning as they are intended and that safe, quality products are produced.
An “audit action report” is used by Specialty Brands to document deviations found during internal self-assessments and during third-party audits. More importantly, it is used to document actions to proactively correct the deficiencies so they are not discovered during the third-party audit. When the auditor delivers the report to the plant, the plant should assemble the management team to review the report point-by-point. A detailed plan of action, including timelines and the person(s) responsible for implementation, should be developed for each element for which the auditor noted a deficiency. The team also should review any comments or recommendations for improvement and consider whether these merit consideration for implementation at the plant. A written response should be prepared from this meeting and sent back to the auditor, and/or to the appropriate customer requesting the audit, to demonstrate the details of the plant’s corrective actions. We have instructed our plants to prepare the audit action report within two weeks after receipt of the final audit report. The report format should include columns for reporting the location, date and section number, the issue/findings, department and party responsible, due date, and corrective actions completed.
Audit Process Practice Really Does Make Perfect
The plant uses the action plan to establish corrective actions and as documentation that these actions have been completed. There are few things more frustrating to corporate officers, such as myself, than to see the same deficiency repeated on an audit from one year to the next. I will also refer to this plan when I visit the plants to verify that they are making progress on corrective actions. In addition, I use the information from the audit to establish justification for plant capital improvement projects.
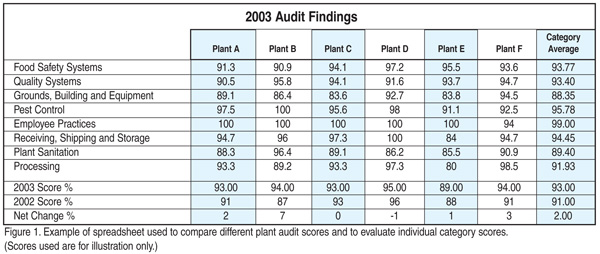 I have found that it is valuable to take the audits from each of the plants and create a spreadsheet to compare plant scores, as well as to evaluate individual category scores (Figure 1). I can use this information, coupled with information from the audit comment sections, to determine what Specialty Brands’ food safety and quality procedures need to be addressed for improvement or where training may be necessary to assure effective implementation of the procedures. It is also worth noting that these types of spreadsheets can be easily attached to new specialty audit software packages that assist in automating your audit forms, records and score sheets. Automating the audit process makes it easier to track the auditing process and corrective actions.
I have found that it is valuable to take the audits from each of the plants and create a spreadsheet to compare plant scores, as well as to evaluate individual category scores (Figure 1). I can use this information, coupled with information from the audit comment sections, to determine what Specialty Brands’ food safety and quality procedures need to be addressed for improvement or where training may be necessary to assure effective implementation of the procedures. It is also worth noting that these types of spreadsheets can be easily attached to new specialty audit software packages that assist in automating your audit forms, records and score sheets. Automating the audit process makes it easier to track the auditing process and corrective actions.
Although tedious at times, the audit system provides a continual educational process for each facility and management team, which allows continuous improvement. We use all of these tools to improve our company performance to provide our customers and consumers with products that they can rely on for safety and quality. It has helped our company improve on our audit performance, has improved our attitude toward the process and can help you, too, if you use the same approach. It may even provide you with more time to work on that budget process!
Michael M. Cramer is Vice President of Food Safety and Quality Assurance for Specialty Brands, Inc, Ontario, California. He is responsible for development and implementation of food safety and quality programs for the company. He spent 16 years with Armour Swift-Eckrich at both plant and corporate locations in poultry and processed meats quality assurance before joining Specialty Brands in 1993. Cramer has authored and spoken on the topics of biosecurity, sanitation, allergen management and quality programs. He is a director with National Meat Association, serves on the Editorial Advisory Board of Food Safety Magazine and is a professional member of the Institute of Food Technologists.
Tammi Frederick is Vice President of Technical Services with Food Safety Net Services, Ltd., a comprehensive, integrated network of laboratory, consulting, auditing and educational services complemented by crisis management programs and regulatory support. She is responsible for the overall implementation and execution of all technical services, including consulting, auditing, education and research. In this role, Frederick provides food safety, quality and animal welfare expertise to clients in all facets of the industry and brings more than 12 years of experience to her position. Prior to joining Food Safety Net Services, Frederick worked in the beef industry for L&H Packing Co. and Surlean Foods in Technical Services. She also worked in the poultry industry as Quality Assurance Manager for Plantation Foods.
Awesome Audit Attitudes: Jumping Through Hoops Without Grinding Gears




