The U.S. Food and Drug Administra-tion (FDA) strives to use the best available scientific information to optimize our efforts to protect public health. To do this, we must continually develop and use efficient means of collecting, organizing, and sharing information for making regulatory decisions. Risk analysis is an example of a systematic approach to efficiently integrate science and policy. In risk analysis, there is a coordination of the activities associated with risk assessment, risk management and risk communication.
This approach is increasingly used to guide risk management and trade decisions at national and international levels. In consideration of the principles of risk analysis, FDA's Center for Food Safety and Applied Nutrition (CFSAN) recently developed a new approach for initiating and managing the conduct of risk assessments. This approach and criteria used to identify and select priority risk assessments are described below.
Learning from Experience
 Even before the draft Listeria monocytogenes and Vibrio parahaemolyticus risk assessments were issued for pubic comment in January 2001, CFSAN formed an ad hoc working group to capture its experiences conducting complex quantitative microbial risk assessments. The working group discussed and evaluated lessons learned from these experiences, which led to recommendations for improving our conduct and management of future risk assessment projects. These recommendations are summarized in Table 1.
Even before the draft Listeria monocytogenes and Vibrio parahaemolyticus risk assessments were issued for pubic comment in January 2001, CFSAN formed an ad hoc working group to capture its experiences conducting complex quantitative microbial risk assessments. The working group discussed and evaluated lessons learned from these experiences, which led to recommendations for improving our conduct and management of future risk assessment projects. These recommendations are summarized in Table 1.
A key recommendation was to identify and select major risk assessments conducted by CFSAN using a decision-based approach that considers the appropriate use of its resources. For CFSAN's purposes, "major" risk assessments were defined to include those that are:
• Non-routine and complex
• May be qualitative or quantitative
• Cross-cutting in nature (involve multiple products or the regulatory responsibilities of multiple programmatic offices in CFSAN or among other agencies)
• Require a commitment of significant resources to complete
A selection process is not needed for routine safety/risk assessments such as those conducted in response to food additive petitions.
What is a decision-based approach? First, the process must allow step-wise evaluations of candidate projects with stopping points at appropriate times. Second, the criteria used to evaluate the projects must be clear and related to our need for risk management decisions. Finally, the process must account for available resources versus those resources needed to successfully complete a proposed project. Our most valuable resource is the expertise of our staff. Other resources include financial capability to fund data collection and purchase computers, software systems and other equipment to improve the efficiency of protecting the food supply. Our available resources are limited by the need for timely decisions and competing priorities for our staff's expertise.
Identification and Selection: A Four-Phase Process
Because risk assessments are time-sensitive and resource-intensive, the goal of the selection process is to identify those critical regulatory concerns that would benefit from a risk assessment. The process reflects the need to determine specific risk management and risk assessment questions to be answered, and conduct an evaluation of the feasibility for successfully completing the risk assessment in a timely manner.
 The purpose of this approach is to ensure that the candidate risk assessments will be useful to CFSAN's regulatory programs, are systematically developed based on those regulatory needs and that adequate resources are available to complete the assessment within the required time frame. These four phases of the process are summarized in Table 2.
The purpose of this approach is to ensure that the candidate risk assessments will be useful to CFSAN's regulatory programs, are systematically developed based on those regulatory needs and that adequate resources are available to complete the assessment within the required time frame. These four phases of the process are summarized in Table 2.
Phase 1. Concept Generation. First, we identify risk management problems that a risk assessment would help solve. This includes understanding the nature of the risk such as the characteristics of the hazard, the urgency of the situation and the regulatory options. In this phase, a justification for each candidate risk assessment suggested by CFSAN's program offices is prepared and includes the following information:
• Purpose of the risk assessment
• Scope of the risk management problem
• Importance to CFSAN
• How the risk assessment results would be used by CFSAN
An important aspect of the scope of the project is the articulation of the risk management and risk assessment questions. The questions that the risk managers must answer are different from those that risk assessors should be asked. An example of a risk management problem is: "How should FDA manage the risk of contracting listeriosis from eating ready-to-eat (RTE) foods served in restaurants?" Based on this example of a risk management problem, the questions posed to the risk assessor might include:
• "What is the exposure to Listeria monocytogenes from consuming RTE foods in restaurants?"
• "What is the likelihood of the general population (or a sensitive subset of the population) contracting listeriosis from eating RTE foods in restaurants?"
• "How much is the risk reduced if FDA required 'use by dates' on RTE foods?"
Phase 2. Problem Identification. Next, the candidate risk assessment projects are reviewed and evaluated by CFSAN's senior management. With the assistance of the risk assessment staff, a recommendation is made as to which candidate projects should be conducted or further evaluated. In this phase, the evaluation criteria are focused on the technical merit and regulatory need. Example recommendations include: (1) The candidate risk assessment should be conducted; (2) the candidate risk assessment has merit but the feasibility of conducting the risk assessment should be evaluated (see Phase 3); or (3) the candidate risk assessment should not be conducted either because it does not fall within CFSAN's regulatory priorities at the current time or it is not needed to make a regulatory decision. Another reason not to conduct a risk assessment is that an immediate decision is needed and insufficient time would be available to complete a risk assessment.
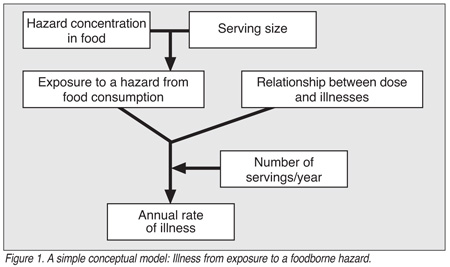 Phase 3. Data Feasibility Evaluation. The goal of this phase is to determine the availability and quality of the data needed to answer the risk assessment questions. This includes determining the available data and the uncertainty created by the lack of data. During this phase, a conceptual model will be generally developed to illustrate the anticipated data inputs, flow of calculations and model outputs. A simple conceptual model is shown in Figure 1.
Phase 3. Data Feasibility Evaluation. The goal of this phase is to determine the availability and quality of the data needed to answer the risk assessment questions. This includes determining the available data and the uncertainty created by the lack of data. During this phase, a conceptual model will be generally developed to illustrate the anticipated data inputs, flow of calculations and model outputs. A simple conceptual model is shown in Figure 1.
Examples of recommendations that could arise from this analysis include:
• The available data will support the conduct of a quantitative risk assessment
• Insufficient data are available for a quantitative risk assessment; conduct a qualitative risk assessment
• Additional research is needed before a risk assessment can be conducted
• The posed risk assessment/risk management questions cannot be answered; modified questions would allow conduct of an alternative risk assessment
Phase 4. Disposition. In the last phase of the identification and selection process, CFSAN's senior management makes a decision for each candidate risk assessment. Factors relied upon to make these decisions include the technical merit, regulatory needs, time frame for making the risk management decision, available staff, available data and other factors including public perception.
Example recommendations include (1) that data and resources are available and CFSAN's regulatory program would benefit, so conduct the risk assessment and assign staff; or (2) that either data or resources are currently inadequate, and thus retain the risk assessment on a list of potential risk assessments pending acquisition of needed data or availability of resources.
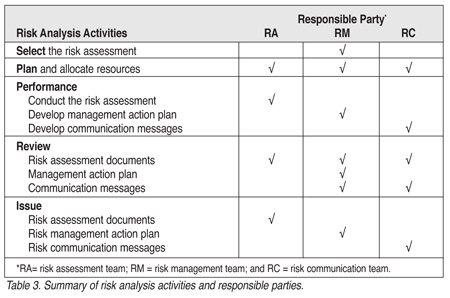 Upon commissioning the selected risk assessment, a series of activities are initiated as summarized in Table 3. The risk assessment, risk management and risk communication teams are responsible for specific tasks in planning, performing, reviewing and issuing documents, action plans and other communications. These activities are discussed in detail in the working group's document titled, "Initiation and Conduct of All 'Major' Risk Assessments within a Risk Analysis Framework," which is available at www.cfsan.fda.gov/~dms/rafw-toc.html.
Upon commissioning the selected risk assessment, a series of activities are initiated as summarized in Table 3. The risk assessment, risk management and risk communication teams are responsible for specific tasks in planning, performing, reviewing and issuing documents, action plans and other communications. These activities are discussed in detail in the working group's document titled, "Initiation and Conduct of All 'Major' Risk Assessments within a Risk Analysis Framework," which is available at www.cfsan.fda.gov/~dms/rafw-toc.html.
Stakeholder Involvement
CFSAN is committed to involving our stakeholders in our activities. Each year, CFSAN holds a public meeting to discuss the achievements of the previous year and present the new program priorities. During and following this meeting, the public is invited to comment on our plans. Once a risk assessment is selected, CFSAN publishes a request for input from stakeholders in the Federal Register. Additional public meetings are typically held throughout the process of conducting the risk assessment. An excellent resource for information on food safety risk assessment is the Joint Institute for Food Safety and Applied Nutrition (JIFSAN) Food Risk Analysis Clearinghouse (www.foodriskclearinghouse.umd.edu).
Conclusion
Developing a process is not the end; it is only the beginning. Once a process is agreed to, it must be clearly communicated and implemented. Next, the process must be followed. To the extent that it is followed, we must ask ourselves the question: "Did it work?" CFSAN is beginning to implement this new selection process. After testing, we will determine whether it facilitated and improved our decision-making and as needed, modify the procedure.
S.B. Dennis is the Risk Assessment Coordinator for the Office of Science, FDA/CFSAN. She is the project manager for the Listeria monocytogenes risk assessment and is actively involved in CFSAN's extramural research activities associated with risk assessment.
The FDA CFSAN Risk Analysis Working Group members included: Robert Buchanan (co-chair), Janice Oliver (co-chair), Philip Bolger, Karen Carson, Marjorie Davidson, Sherri Dennis, Patricia Hansen, Kathy Gombas, Donald Kraemer, John Kvenberg, Wesley Long, Marianne Miliotis, Arthur Miller, Patricia Schwartz, Philip Spiller, Terry Troxell, Richard Whiting and Richard Williams.
Acknowledgments
The authors would like to acknowldege the contribution of Dr. Susan Santos with Focus Group Risk Communication and Environ-mental Management Consultants, who facilitated the CFSAN Risk Analysis Working Group meetings. The authors also thank Dr. Clark Carrington, who assisted in the development of the conceptual model provided in this article.



