Microbiological
Microbiological contamination of food encompasses bacterial pathogens, such as Listeria monocytogenes, Escherichia coli (E.coli), Salmonella, Cronobacter, and many other pathogens that can contaminate food at any point during the supply chain, causing foodborne illness. This category also includes foodborne parasites.
ARTICLES
Medically Important Foodborne Parasites: A Consequential Challenge for Food Safety Assurance
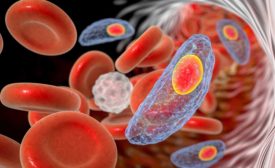
Foodborne parasitic diseases are often overlooked in food safety control schemes, even though they are known to pose a severe threat to human health
April 11, 2024
Get our eMagazine delivered directly to your inbox
Stay in the know on the latest science-based solutions for food safety.
SUBSCRIBE TODAY!Copyright ©2024. All Rights Reserved BNP Media.
Design, CMS, Hosting & Web Development :: ePublishing